Project CBD: CBD and Parkinson’s Disease
The endocannabinoid system and digestive imbalance play major roles in Parkinson’s disease. Research on CBD, THC, and THCV has demonstrated that cannabis medicine may help to manage PD symptoms.
- The endocannabinoid system plays a major role in Parkinson’s Disease (PD).
- PD is associated with impairment of motor control after the loss of 60-80% of dopamine-producing neurons in the brain region.
- Digestive imbalance may play a role in the advancement of PD & the severity of symptoms.
- Cannabinoids have neuroprotectant, anti-oxidant and anti-inflammatory properties which may be beneficial for managing PD.
- Various combinations of CBD, THC, and THCV may provide relief for Parkinson’s symptoms.

Scientists at the University of Louisville School of Medicine in Kentucky have identified a previously unknown molecular target of cannabidiol (CBD), which may have significant therapeutic implications for Parkinson’s Disease (PD).
A poster by Zhao-Hui Song and Alyssa S. Laun at the 2017 meeting of the International Cannabinoid Research Society in Montreal disclosed that CBD activates a G-coupled protein receptor called “GPR6” that is highly expressed in the basal ganglia region of the brain. GPR6 is considered an “orphan receptor” because researchers have yet to find the primary endogenous compound that binds to this receptor.(1)
It has been shown that a depletion of GPR6 causes an increase of dopamine, a critical neurotransmitter, in the brain. This finding suggests GPR6 could have a role in the treatment of Parkinson’s, a chronic, neurodegenerative disease that entails the progressive loss of dopaminergic (dopamine-producing) neurons and consequent impairment of motor control.
By acting as an “inverse agonist” at the GPR6receptor, CBD boosts dopamine levels in preclinical studies.
Parkinson’s affects an estimated 10 million people worldwide, including one million Americans. It is the second most common neurological disorder (after Alzheimer’s Disease). Over 96 percent of those diagnosed with PD are over 50 years old with men being one-and-a-half times more likely to have PDthan women. Uncontrolled PD significantly reduces the patient’s quality of life and can render a person unable to care for themselves, trapped in a body they cannot control.
Dopamine depletion
Parkinson’s Disease is most associated with compromised motor function after the loss of 60-80% of dopamine-producing neurons. As dopaminergic neurons become damaged or die and the brain is less able to produce adequate amounts of dopamine, patients may experience any one or combination of these classic PD motor symptoms: tremor of the hands, arms, legs or jaw; muscle rigidity or stiffness of the limbs and trunk; slowness of movement (bradykinesia); and /or impaired balance and coordination (postural instability).
Additional symptoms include decreased facial expressions, dementia or confusion, fatigue, sleep disturbances, depression, constipation, cognitive changes, fear, anxiety, and urinary problems. Pesticide exposure and traumatic brain injury are linked to increased risk for PD. Paraquat, an herbicide sprayed by the DEA in anti-marijuana defoliant operations in the United States and other countries, resembles a toxicant MPTP [methyl-phenyl-tetrahydropyridien], which is used to simulate animal models of Parkinson’s for research purposes.(2)
Within the PD brain there are an inordinate number of Lewy bodies – intracellular aggregates of difficult to break down protein clusters – that cause dysfunction and demise of neurons.(3) This pathological process results in difficulties with thinking, movement, mood and behavior. The excessive presence of Lewy bodies, coupled with the deterioration of dopaminergic neurons, are considered to be hallmarks of Parkinson’s. But mounting evidence suggests that these aberrations are actually advanced-stage manifestations of a slowly evolving pathology.
It appears that non-motor symptoms occur for years before the disease progresses to the brain, and that PD is actually a multi-system disorder, not just a neurological ailment, which develops over a long period of time. According to the National Parkinson’s Foundation, motor symptoms of PD only begin to manifest when most of the brain’s dopamine-producing cells are already damaged.
Patients whose PD is diagnosed at an early stage have a better chance of slowing disease progression. The most common approach to treating PD is with oral intake of L-dopa, the chemical precursor to dopamine. But in some patients, long-term use of L-dopa will exacerbate PD symptoms. Unfortunately, there is no cure – yet.

Gut-brain axis
What causes Parkinson’s?
One theory that is gaining favor among medical scientists traces the earliest signs of PD to the enteric nervous system (the gut), the medulla (the brainstem), and the olfactory bulb in the brain, which controls one’s sense of smell. New research shows that the quality of bacteria in the gut – the microbiome – is strongly implicated in the advancement of Parkinson’s, the severity of symptoms, and related mitochondrial dysfunction.
Defined as “the collection of all the microorganisms living in association with the human body,” the microbiome consists of “a variety of microorganisms including eukaryotes, archaea, bacteria and viruses.” Bacteria, both good and bad, influence mood, gut motility, and brain health. There is a strong connection between the microbiome and the endocannabinoid system: Gut microbiota modulate intestinal endocannabinoid tone, and endocannabinoid signaling mediates communication between the central and the enteric nervous systems, which comprise the gut-brain axis.
Viewed as “the second brain,” the enteric nervous system consists of a mesh-like web of neurons that covers the lining of the digestive tract – from mouth to anus and everything in between. The enteric nervous system generates neurotransmitters and nutrients, sends signals to the brain, and regulates gastrointestinal activity. It also plays a major role in inflammation.
The mix of microorganisms that inhabit the gut and the integrity of the gut lining are fundamental to overall health and the ability of the gut-brain axis to function properly. If the lining of the gut is weak or unhealthy, it becomes more permeable and allows things to get into the blood supply that should not be there, negatively impacting the immune system. This is referred to as “leaky gut.” Factor in an overgrowth of harmful bacteria and a paucity of beneficial bacteria and you have a recipe for a health disaster.
The importance of a beneficial bacteria in the gut and a well-balanced microbiome cannot be overstated. Bacterial overgrowth in the small intestine, for example, has been associated with worsening PD motor function. In a 2017 article in the European Journal of Pharmacology, titled “The gut-brain axis in Parkinson’s disease: Possibilities for food-based therapies,” Peres-Pardo et al examine the interplay between gut dysbiosis and Parkinson’s. The authors note that “PD pathogenesis may be caused or exacerbated by dysbiotic microbiota-induced inflammatory responses … in the intestine and the brain.”(4)
Mitochondria, microbiota and marijuana
The microbiome also plays an important role in the health of our mitochondria, which are present in every cell in the brain and body (except red blood cells). Mitochondria function not only as the cell’s power plant; they also are involved in regulating cell repair and cell death. Dysfunction of the mitochondria, resulting in high levels of oxidative stress, is intrinsic to PD neurodegeneration. Microbes produce inflammatory chemicals in the gut that seep into the bloodstream and damage mitochondria, contributing to disease pathogenesis not only in PD but many neurological and metabolic disorders, including obesity, type-2 diabetes, and Alzheimer’s.
The evidence that gut dysbiosis can foster the development of PD raises the possibility that those with the disease could benefit by manipulating their intestinal bacteria and improving their microbiome. Enhancing one’s diet with fermented foods and probiotic supplements may improve gut health and relieve constipation, while also reducing anxiety, depression and memory problems that afflict PD patients.
Cannabis therapeutics may also help to manage PD symptoms and slow the progression of the disease. Acclaimed neurologist Sir William Gowers was the first to mention cannabis as a treatment for tremors in 1888. In his Manual of Diseases of the Nervous System, Grower noted that oral consumption of an “Indian hemp” extract quieted tremors temporarily, and after a year of chronic use the patient’s tremors nearly ceased.
Modern scientific research supports the notion that cannabis could be beneficial in reducing inflammation and assuaging symptoms of PD, as well as mitigating disease progression to a degree. Federally-funded preclinical probes, for example, have documented the robust antioxidant and neuroprotective properties of CBD and THC with “particular application … in the treatment of neurodegenerative diseases, such as Alzheimer’s disease, Parkinson’s disease and HIV dementia.” Published in 1998, these findings formed the basis of a U.S. government patent on cannabinoids as antioxidants and neuroprotectants.
Pot for Parkinson’s
Although clinical studies focusing specifically on the use of plant cannabinoids to treat PD are limited (because of marijuana prohibition) and convey conflicting results, in aggregate they provide insight into how cannabis may aid those with Parkinson’s. Cannabidiol, THC, and especially THCV all showed sufficient therapeutic promise for PD in preclinical studies to warrant further investigation. Additional research might shed light on which plant cannabinoids, or combination thereof, is most appropriate for different stages of Parkinson’s.
Anecdotal accounts from PD patients using artisanal cannabis preparations indicate that cannabinoid acids (present in unheated whole plant cannabis products) may reduce PD tremor and other motor symptoms. Raw cannabinoid acids (such as CBDA and THCA) are the chemical precursors to neutral, “activated” cannabinoids (CBD, THC). Cannabinoid acids become neutral cannabinoid compounds through a process called decarboxylation, where they lose their carboxyl group through aging or heat. Minimal research has focused on cannabinoid acids, but the evidence thus far confirms that they have powerful therapeutic attributes, including anti-inflammatory, anti-nausea, anti-cancer, and anti-seizure properties. In a 2004 survey of cannabis use among patients at the Prague Movement Disorder Centre, 45 percent of respondents reported improvement in PD motor symptoms.

Cannabis clinicians are finding that dosage regimens for medical marijuana patients with PDdon’t conform to a one-size-fits-all approach. In her book Cannabis Revealed (2016), Dr. Bonni Goldstein discussed how varied a PD patient’s response to cannabis and cannabis therapeutics can be:
“A number of my patients with PD have reported the benefits of using different methods of delivery and different cannabinoid profiles. Some patients have found relief of tremors with inhaled THC and other have not. A few patients have found relief with high doses of CBD-rich cannabis taken sublingually. Some patients are using a combination of CBD and THC … Trial and error is needed to find what cannabinoid profile and method will work best. Starting a low-dose and titrating up is recommended, particularly with THC-rich cannabis. Unfortunately, THCV-rich varieties are not readily available.”
Juan Sanchez-Ramos M.D., PhD, a leader in the field of movement disorders and the Medical Director for the Parkinson Research Foundation, told Project CBD that he encourages his patients to begin with a 1:1 THC:CBD ratio product if they can get it. In a book chapter on “Cannabinoids for the Treatment of Movement Disorders,” he and coauthor Briony Catlow, PhD, describe the dosage protocol used for various research studies that provided statistically positive results and a dosing baseline for PD. This data was included in a summary of dosing regimens from various studies compiled by Dr. Ethan Russo:
- 300 mg/day of CBD significantly improved quality of life but had no positive effect on the Unified Parkinson Disease Rating Scale. (Lotan I, 2014)
- 0.5 g of smoked cannabis resulted in significant improvement in tremor and bradykinesia as well as sleep. (Venderová K, 2004)
- 150 mg of CBD oil titrated up over four weeks resulted in decreased psychotic symptoms. (Chagas MH, 2014)
- 75-300 mg of oral CBD improved REM-behavior sleep disorder. (Zuardi AW, 2009)
Chronic Advice
Of course, each patient is different, and cannabis therapeutics is personalized medicine. Some people find the THC-induced cannabis high to be disagreeable. Generally speaking, an optimal therapeutic combination will include a synergistic mix of THC and CBD – although PD patients with sleep disturbances may benefit from a higher THC ratio at night.
Dr. Russo offers cogent advice for patients with PD and other chronic conditions who are considering cannabis therapy. “In general,” he suggests, “2.5 mg of THC is a threshold dose for most patients without prior tolerance to its effects, while 5 mg is a dose that may be clinically effective at a single administration and is generally acceptable, and 10 mg is a prominent dose, that may be too high for naïve and even some experienced subjects. These figures may be revised upward slightly if the preparation contains significant CBD content … It is always advisable to start at a very low dose and titrate upwards slowly.”
For information about nutritional supplementation to help manage PD, visit the Life Extension Foundation Parkinson’s page.
Lifestyle Modifications for PD Patients
It is important to treat the patient as a whole – mind, body and soul. The following are a few lifestyle modifications that may provide relief from PD symptoms and improve quality of life.
- Do cardio aerobic exercise: This benefits the body in so many ways, including stimulating the production of one’s endocannabinoids, increasing oxygen in the blood supply, mitigating the negative impact of oxidative stress, and boosting the production of BDNF, a brain-protecting chemical found to be low in PD patients.
- Eat more fruits and vegetables: The old saying “garbage in, garbage out” is so true. The majority of PD patients suffer from chronic constipation. A high fiber diet can be helpful in improving gut motility and facilitate daily bowel movements.
- Get restful sleep: Not getting good sleep plays a major factor in one’s immune function, cognition and quality of life. The importance of adequate restful sleep cannot be over emphasized.
- Reduce protein intake – This may help reduce the accumulation of protein bodies that result in Lewy bodies that appear in the enteric nervous system and the central nervous system and increase the uptake of L-dopa.
- Practice meditation, yoga or Tai Chi: The focus on the integration of movement and breath not only improve mobility but it also improves cognition and immunity. One study showed an increase in grey matter density in the areas of the brain associated with PD. Another showed that yoga improved balance, flexibility, posture and gait in PD patients. Research shows that tai chi help improve balance, gait, functional mobility and overall well being.
- Consume probiotic food and supplements: Probiotic foods — raw garlic, raw onions, bananas, asparagus yams, etc.— are a great source for the good bacteria in your large intestine. Augmenting your diet with probiotic supplements, especially after taking antibiotics, can support the immune system by helping to repopulate the upper digestive tract with beneficial bacteria. Consult your doctor for a recommendation for a quality probiotic.
- Drink coffee: The risk of PD is considerably lower for men who consume coffee daily.
Nishi Whiteley, a Project CBD research associate and contributing writer, is the author of Chronic Relief: A Guide to Cannabis for the Terminally and Chronically Ill (2016). Special thanks to Juan Sanchez-Ramos for reviewing this article, Ethan B. Russo, M.D. for providing a summary of Parkinson’s research for inclusion in this article, and to Adrian Devitt-Lee for his research support.
Sources:
- Abrams, D. (2010, Winter). Cannabis in Pain and Palliative Care. The Pain Practitioner, pp. 35-45.
- AC Howlett, F. B. (2002). International Union of Pharmacology. XXVII. Classification of cannabionid receptros. Pharmacological Reviews, 161-202.
- Aidan J. Hampson, J. A. (2003). USA Patent No. 6,630,507.
- Barbara A. Pickut, W. V. (2013). Mindfulness based intervention in Parkinson’s disease leads to structural brain changes on MRI A randomized longitudinal study. Clinical Neurology and Neurosurgery, 2419-2425.
- Birony Catlow, J. S.-R. (2015). Cannabinoids for the Treatment of Movement Disorders. Current Treatment Options in Neurology.
- C Garcia, C. P.-G.-A.-R. (2011). Symptom-relieving and neuroprotective effects of the phytocannabinoid Δ9-THCV in animal models of Parkinson’s disease. British Journal of Pharmacology, 1495-1506.
- Chagas MH, Z. A.-P. (2014). Effects of cannabidiol in the treatment of patients with Parkinson’s disease: an exploratory double-blind trial. Journal of Phsychopharmacology, 1088-98.
- David N. Hauser, T. G. (2013). Mitochondrial dysfunction and oxidative stress in Parkinson’s disease and monogenic parkinsonism. Neurobiology of Disease, 35-42.
- David Perlmutter, M. (2015). Belly and Brain on Fire. In M. David Perlmutter, Brain Maker (pp. 49-70). New York: Little, Brown and Company.
- Foundation, N. P. (2017, June 19). What is Parkinson’s. Retrieved from National Parkinson’s Foundation: http://www.parkinson.org/understanding-parkinsons/what-is-parkinsons
- Foundation, P. D. (2017, 6 21). Parkinson’s Statistics. Retrieved from Parkinson’s Disease Foundation: http://www.pdf.org/parkinson_statistics
- Goldstein, B. (2016). Parkinson’s Diseas. In B. Goldstein, Cannabis Revealed (pp. 206-208). Bonni Goldstein.
- L. Klingelhoefer, H. R. (2017). Hypothesis of Ascension in Idiopathic Parkinson’s Disease. Neurology Intereatnional,E28-35.
- Leonard L. Sokol, M. J. (2016). Letter to the Editor: Cautionary optimism: caffeine and Parkinson’s disease risk. Journal of Clinical Movement Disorders, pp. 3-7.
- Lisa Klingelhoefer, H. R. (2015). Pahtogenesis of Parkinson disease—the gut-brain axis and environmental factors. Nature, 625-636.
- Lotan I, T. T. (2014). Cannabis (medical marijuana) treatment for motor and non-motor symptoms of Parkinson disease:. Clin Neuropharmacol, 37(2)-41-4.
- Madeleine E. Hackney1 and Gammon M. Earhart1, 2. (2008). Tai Chi Improves Balance and Mobility in People with Parkinson Disease. Gait and Posture, 456-460.
- National Institute of Health. (2017). NIH Human Microbiome Project HOME Page. Retrieved from NIH Human Microbiome Project: http://hmpdacc.org/overview/about.php
- Pal Pacher, S. B. (2006). The Endocannabinoid System as an Emerging Target. Pharmacological Reviews, 389-462.
- Parker, R. M. (2013). The Endocannabinoid System and the Brain. The annual Review of Psychology, 21-47.
- Paula Perez-Pardo, T. K. (2017). The gut-brain axis in Parkinson’s disease: Possibilities for food -based therapies. European Journal of Pharmacology, http://www.sciencedirect.com/science/article/pii/S0014299917303734.
- Perlmutter, D. (2015). Brain Maker. New York: Little, Brown and Company.
- Russo, E. (2011). Taming THC: potential cannabis synergy and phytocannabinoid-terpenoid entourage effects. British Journal of Pharmacology, 1344-64.
- Russo, E. B. (2015). Current status and future of cannabis research. Clinical Researcher, 58-63.
- Russo, E. B. (2015, January). Introduction to the Endocannabinoid system. Retrieved from Phytecs.com: http://www.phytecs.com/wp-content/uploads/2015/02/Russo-Introduction-to-…
- Russo, E. B. (2016). The Medical Use of Cannabis and Cannabinoids in Parkinson’s Disease. Retrieved from The Answer Page: https://www.theanswerpage.com/
- Schecter, G. L. (2010). The Endocannabniond system. In J. Holland, The Pot Book (pp. 52-62). Rochester, Vermont: Park Street Press.
- Venderová K, R. E. (2004). Survey on cannabis use in Parkinson’s disease: subjective improvement of motor symptoms. Movement Disorder, 1102-6.
- Yevonne Searls Carlgrove, N. S. (2012). Effect of Yoga on Motor Function in People with parkinosn’s Disease: A Randomized Controled Piolot Study. Yoga and Physical Therapy.
- Yudowski, D. A. (2017). Cannabinoid Receptors in the Central Nervous System: Their Signaling and Roles in Disease. Frontiers in Cellular Nueroscience, article 294.
- Zuardi AW, C. J. (2009). Cannabidiol for the treatment of psychosis in Parkinson’s disease. Journal of Phsychopharmacology, 979-83.
(1) An inverse agonist binds directly to a receptor and modifies it in a way that causes the receptor to have the opposite effects of activating it normally.
(2) MPTP was found in an underground meperidine (Demerol) synthesis that caused a small epidemic of Parkinson syndrome in i.v. drug abusers in the San Francisco area in the mid-1980s.
The presence of Lewy bodies (a-synuclein protein clusters) in other parts of the body could potentially serve as an early detection marker for PD, especially in the olfactory bulb and the enteric nervous system.
(4) Peres-Prado et al analyzed gut microbiota in PD patients compared to controls and found the following:
- Prevotellaceae, a bacterium which supports the production of health-promoting short chain fatty acids (SCFA), biosynthesis of thiamine and folate, and is thought to be associated with increased gut permeability, was 78% lower in the feces of PD patients versus that of their sex-matched and age-matched controls.
- Biopsies of colonic tissue retrieved from PD patients indicate high levels of tumor necrosis factor-alpha and other inflammatory agents.
- A lower abundance of SCFA-producing and anti-inflammatory bacteria from the class of Blautia, Coprococcus, and Roseburia were found in fecal samples of PD patients. (Paula Perez-Pardo, 2017)
- Gastric abnormalities may increase small intestinal bacterial overgrowth (SIBO). SIBO is prevalent in PD patients and correlates directly to worse motor dysfunction.
- Gut-derived lipopolysaccharide (LPS – an inflammatory toxin produce by bacteria) promotes the disruption of the blood-brain barrier.
- Impaired gherlin, a gut hormone known as the hunger hormone, is thought to be associated with maintenance and protection of dopamine function in the nigrostriatal pathway which is one of four major dopamine pathways and is particularly involved in movement. Impaired gherlin has been reported in PD patients.
Photo credits: Pixabay, Ispectrum Magazine.
_______________________________________________________________________________________________
ALTHealth Works: Young Cancer Patient Already Planned His Own Funeral – You Won’t Believe What Happened Just 5 Days After a Controversial Treatment…
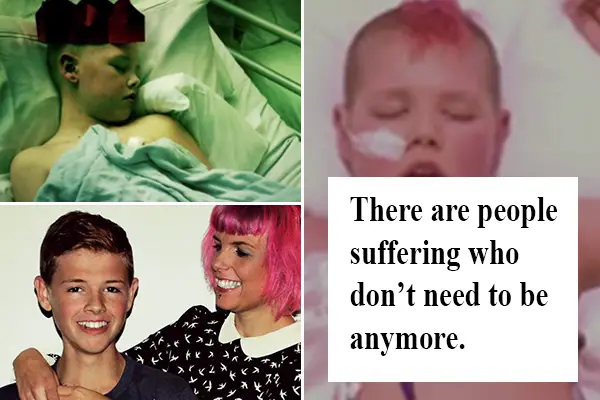
Some forces in the world are so strong that they win over fear and pain.
Such is the tremendous strength of a mother when protecting her children.
When Carrie Blackwell’s son Deryn exhausted all options of modern medicine, she risked spending 14 years behind bars, when giving him cannabis in an attempt to at least alleviate his symptoms.
This risky choice not only removed Deryn’s suffering, but also may have become the missing puzzle piece that led to his recovery.
This recovery was astonishing as Deryn’s health turned from serious to life-threatening.
At the age of 10, Deryn was diagnosed with leukemia, and two years later he developed an extremely rare cancer called Langerhans cell sarcoma, which appeared in his tonsils. Langerhans cell sarcoma has only been reported in English literature a total of 37 times, and Deryn is one of five people said to have had it in the world today.
Not only did he have this rare cancer, he is also the said to be the only boy in the world to have these two cancers at the same time. For this reason, he has been called “The Boy in 7 Billion,” which is also the name of Carrie’s book, which tells Deryn’s story in full detail.
Deryn has four unsuccessful bone marrow transplants, which left his immune system completely shut down. He was surviving merely on antibiotics. And after a four-year battle, at the age of 14, he was placed into a children’s hospice.
“Standing in the dimly lit bedroom in the hospice looking over the body of my sleeping eldest son, I couldn’t hold back the words, or emotions, any longer. As tears rolled down my face, my thoughts came tumbling out loud… ‘This isn’t fair, this isn’t right. You’re only 14 years old,’” writes Carrie in her book.
At this time, Deryn was only given a few days left to live. Yet, he was in such an unimaginable pain and suffering that he wanted to and attempted to kill himself. He even begged his mom to take him to Brussels, because he heard that there, it is legal to use euthanasia for children over the age of 12.
“It sounds awful, but he looked forward to dying because after being ill for four years, he just wanted an end to it,” Carrie said.
Feeling desperate, she did something she was absolutely hesitant to do for a long time – she gave her son a cannabis tincture in hopes that it would relieve his pain at least a little bit. She had no hopes of her son surviving, as he had already planned his funeral. She also knew was she did was illegal in the U.K.
“I was willing to face 14 years, every single day of 14-year sentence, just to give him the alleviation that it gave him,” she said.
Cannabis worked immediately – Deryn felt relaxed, and no longer anxious or in pain. For his mom, that is more than she could have ever asked for. But something remarkable, something completely unexpected has also happened. After five days of cannabis, Deryn’s body began producing white blood cells needed for the body to heal. At day 104, he felt completely well.
“Honestly the effects of it blew my mind. It wasn’t what I expected,” Carrie told This Morning show.
She asked the doctor if it could be any drugs that they gave him and was told – no. She says she even tested if it was the cannabis by stopping this treatment, and the white blood count fell again.
Carrie does not believe that cannabis cured Deryn, yet she finds no answer, other than it was cannabis that provided the final and needed push needed for Deryn’s recovery.
Some call this recovery a “medical miracle.” Many do not believe in miracles, and yet, Deryn today is alive, out of the hospital, and is about to celebrate his 18th birthday at the end of the year.
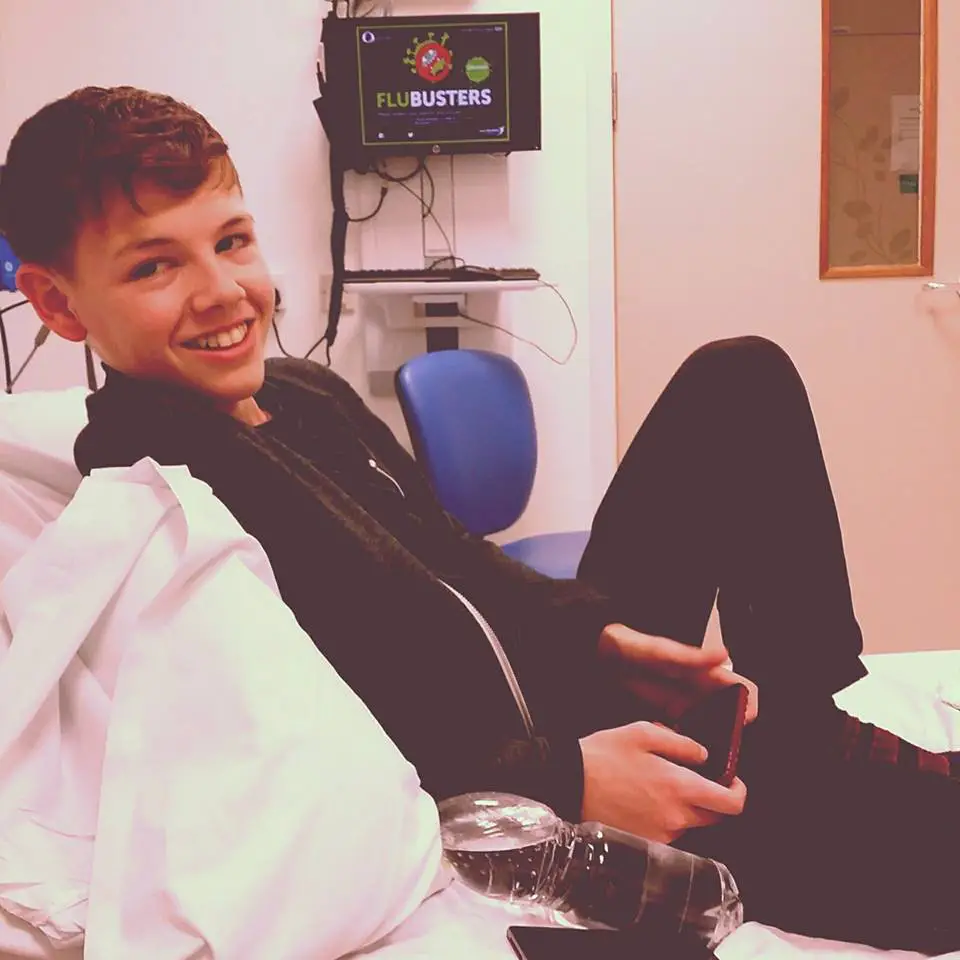
It’s not the end of the journey for Deryn as the radiation he had for his cancers can cause other cancers. So far, Carrie has been able to treat some long term side effects with alternative methods. PHOTO: Facebook/CallieBlackwellAuthor
Did Medical Cannabis Research Become a Black and White Issue?
There are many articles covering this story calling this as a direct proof that cannabis cures cancer. And some commenters get upset that this story is being represented as a cannabis-cure story. In reality, we do not know that for sure – not yet. Science has not proven cannabis to “cure” cancer, but it is important to recognize that it also has not proven the opposite – it has not proven that cannabis does not cure cancer, and it has shown many beneficial effects for those who suffer.
Many studies, even quoted by the American Cancer Organization, has shown its powerful ability to kill cancer cells in the lab: cannabinoids (active compounds in marijuana) prevented tumor growth and promoted cancer cell death in rats and mice, reduced the risk of colon cancer in mice, killed liver cancer cells, and made chemotherapy more effective.
Besides killing cancer cells, studies have also shown cannabis to be useful for stimulating appetite (a real issue for cancer patients), relieving pain, controlling nausea caused by chemo, and calming anxiety.
Thoughts on The Double Standard Between Chemo and Cannabis
Personally, I believe that there is an unfair double standard in the modern-versus-alternative medicine conversation.
Chemotherapy, the most commonly-used cancer treatment today, is not a magic bullet. Even the Cancer Society does not claim that it kills cancer cells, instead phrasing it as ‘it helps to kills cancer cells.’ But when a powerful herb or a supplement helped to heal someone from cancer, suddenly it does not count, because it is said that it did not cure it alone.
Chemo drugs are rarely given separately; a few if not a dozen are usually needed to treat a patient. Yet, when it comes to cannabis, it sometimes becomes a black and white issue – it either cures cancer does not. What if the truth is somewhere in the middle: what if it helps when used in combination with other treatments, either conventional or alternative, but helps so greatly, that is may be a difference between life and death? Meanwhile, unlike chemo, cannabis’s tiny side effects cannot hurt or kill the patient.
Deryn’s mom herself keeps repeating that she does never claim and does not believe that cannabis cured Deryn. However, she wants the medical establishment to do more research to find out what medical properties cannabis does have. After all, it jump started Daryn’s immune system, and if it was not for cannabis, he might not have been alive today. Besides, the way it alleviated Daryn’s suffering may be priceless. And for the Blackwell family, that made all the difference.
Today, Carrie is an active advocate for medical cannabis research. Just recently, she traveled to Portugal to meet with the pioneers of this research. You can follow her mission on her website, and read the emotional story of her son in the book “The Boy in 7 Billion.”
___________________________________________________________
GFarma: The Reason You Get a ‘Runner’s High’ Is Not What You Think
No, it’s not endorphins.
There’s possibly no better feeling than the calm and happiness that follow the completion of a long, tough run.
So where does this so-called ‘runner’s high’ come from?
You’ve probably heard it get chalked up to a rise in endorphins, the ‘happy’ chemicals that induce feelings of pain relief and pleasure. But it’s actually more complicated than that.
The ‘endorphins make you happy’ idea
The idea that increased levels of endorphins are responsible for that post-workout happy feeling came out of 1980s research that showed endorphin levels in the blood spiked after prolonged exercise.
Some researchers assumed these chemicals must also produce the sense of euphoria we feel after a workout.
But recent studies in mice suggest that endorphins actually might not have anything to do with the runner’s high.
The problem with the endorphin explanation is that they’re very large molecules – so large, in fact, that they can’t move from the blood into the brain.
The blood-brain barrier is key to keeping the brain safe, since it stops certain pathogens and molecules from passing from the blood into the brain.
Because endorphins can’t get through, it’s unlikely that they are the sole chemical responsible for the feelings associated with vigorous exercise.
Instead, scientists think the effect can be attributed to other chemicals in the body that produce similar pain-relieving and happy feelings.
Turning to endocannabinoids
Levels of a chemical called anandamide also increase when you exercise, according to a 2015 study in mice and a small 2004 study in people.
Anandamide is a type of endocannabinoid, a chemical that’s part of the system that moderates the psychoactive, feel-good effects of marijuana. And unlike cumbersome endorphins, anandamide can smoothly make its way from the blood to the brain.
For the 2015 paper, researchers at the Central Institute of Mental Health at the University of Heidelberg medical school compared the effects of endorphins and endocannabinoids on mice as they ran on running wheels.
The researchers found that, in addition to appearing calmer and less sensitive to pain after running, the mice had higher levels of both endorphins and endocannabinoids.
The animals also spent more time in well-lit parts of their cage, something calm, less anxious mice tend to do. They were also slightly more pain-tolerant after their stints on the wheel.
To measure the effects of each chemical individually, the researchers gave the mice drugs that blocked the effects of each. When they blocked the endorphins, nothing happened – the animals remained more relaxed and pain-tolerant.
But when they blocked the effects of the endocannabinoids, the symptoms of the mice’s runner’s highs disappeared.
These findings suggest that the mice’s elevated endorphin levels had little to do with their post-workout buzz.
This research has one obvious caveat, however: Mice aren’t humans. And the study also revealed, disappointingly, that you probably need to run pretty far to experience a runner’s high. The mice ran an average of more than three human miles per day (a long way for a mouse).
If you’re interested in learning more about the connection between endocannabinoids and runners’ highs, this video from YouTube channel SciShow walks through some of the evidence.
Other factors at play
Other studies, however, suggest that neither endorphins nor endocannibinoids are the cause of the runner’s high. A 2015 study, for example, found that mice with low levels of a hormone called leptin tended to run farther than mice with normal levels of leptin.
Leptin, otherwise known as the “satiety hormone,” inhibits the feeling of hunger in order to regulate our energy levels.
The idea is that the less full (or more hungry) you feel, the more motivated you are to keep running. And that increased motivation might make it easier to get a runner’s high.
“Ultimately, leptin is sending the brain a clear message: When food is scarce, it’s fun to run to chase some down,” lead study author Maria Fernanda Fernandes told Outside Magazine in 2015.
But again, the fact that these results have been demonstrated in mice doesn’t mean the same effects will necessarily be found in humans.
And because there might be a combination of factors at play, definitive evidence of what exactly causes a runner’s high might continue to elude scientists for a while.

